Adverse events should be reported
Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/.
Adverse events should also be reported to Luye Pharma Ltd. at safety@luyepharma.co.uk.

Seroquel XL® and Seroquel®
(quetiapine)
Seroquel® (quetiapine) is for the treatment of schizophrenia, moderate to severe manic episodes in bipolar disorder, major depressive episodes in bipolar disorder and preventing recurrence of manic or depressed episodes in patients with bipolar disorder who previously responded to quetiapine.
Find out more
Fencino®
(Fentanyl Transdermal Patch)
Fencino® (fentanyl) is a transdermal patch indicated for management of severe chronic pain that requires continuous long-term opioid administration. The Fencino® patch should be replaced every 72 hours.
Find out more
Zeyzelf®
(Twice Weekly Rivastigmine Transdermal Patch)
Zeyzelf® (rivastigmine) is a twice weekly transdermal patch indicated for the symptomatic treatment of mild to moderately severe Alzheimer’s dementia. The Zeyzelf® patch should be replaced twice weekly on fixed days (after four and three days, respectively).
Find out more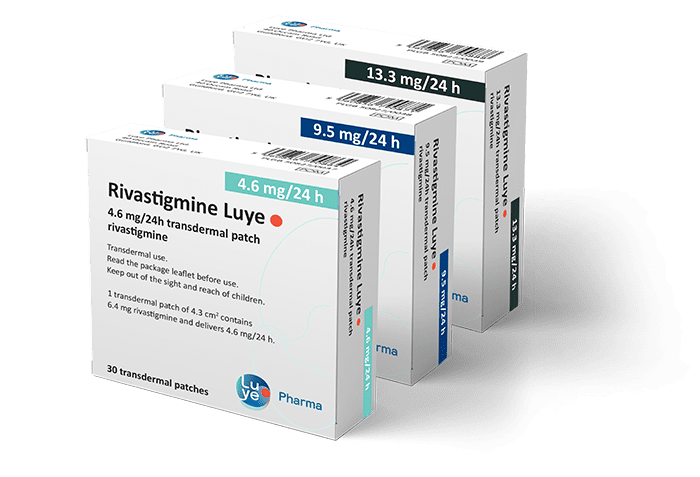
Rivastigmine Luye
(Single Day Rivastigmine Transdermal Patch)
Rivastigmine Luye Single Day Transdermal Patch is used for the treatment of adult patients with mild to moderately severe Alzheimer’s dementia.
Find out more
Rotigotine Luye
(Single Day Rotigotine Transdermal Patch)
Rotigotine Luye is indicated for the treatment of Parkinson’s disease and Restless Legs Syndrome (RLS).
Find out more
Fenylat
(Fentanyl Transdermal Patch)
Fenylat is indicated for management of severe chronic pain. The Fentanyl patch should be replaced every 72 hours.
Find out more